 
When two atoms react to form a compound, it is know as combination reaction. There are main three types of chemical reactions with reference to oxygen and hydrogen: The new substance produced as a result of chemical reaction is called products.įor example: When two sodium atoms react with two chlorine atoms(reactants), they give a completely new compound (product) i.e. Product: the substance which forms as a result of chemical reaction is called products. Reactant: The substance which take part in a chemical reaction are called reactants. S t e p 1 : H 2 O 2 ( a q ) + I - ( a q ) → H 2 O ( l ) + I O - ( a q ) S t e p 2 : I O - ( a q ) + H 2 O 2 ( a q ) → H 2 O ( l ) + O 2 ( g ) + I - ( a q ) The reaction is proceed by two step mechanism: This is called redox reaction.ī.The rate of decomposition of hydrogen peroxide can be increased by having the reaction occurs in the presence of iodide ion. Therefore, in this reaction oxidation of one substance and reduction of another substance take place simultaneously. That is, oxidation of SnCl 2 has taken place. Due to the above reaction the number of chlorine attached to tin atom increases to four. Therefore, reduction of FeCl 3 happened or this reactor on the other hand, before reaction two chlorine atoms where attached with each atom of tin (stannum). That is one negative chlorine atom is released. After the reaction, only two chlorine atoms are attached. 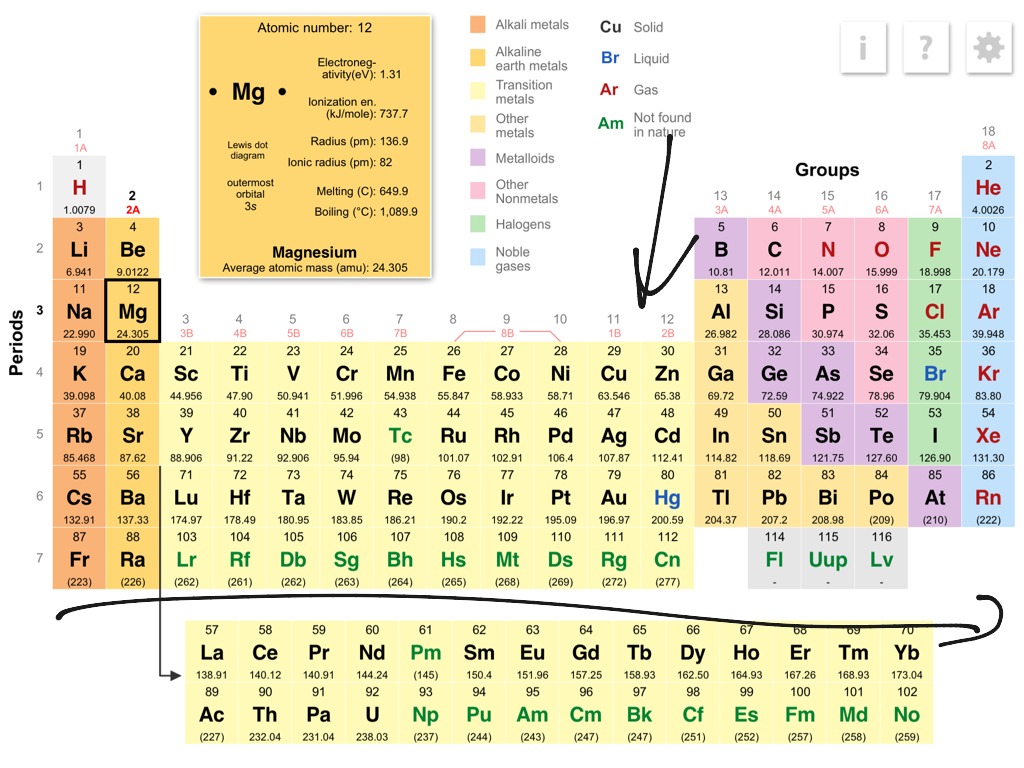
Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.įor example: If we add stannous chloride solution to the yellow solution of ferric chloride then light green ferrous chloride solution and stannic chloride solution are produced.Ģ F e C l 3 ( a q ) + S n C I 2 ( a q ) → 2 F e C l 2 ( a q ) + S n C I 4 ( a q )īefore the reaction, 3Cl atoms were attached to each iron atom.Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion.The process in which oxidation and reduction occurs simutaneously is called as redox reaction. The trends in ionisation energies across periods and down groups can be explained in terms of the atomic size, nuclear charge and the screening effect due to inner shell electrons.C4.1b explain how observed simple properties of Groups 1, 7 and 0 depend on the outer shell of electrons of the atoms and predict properties from given trends down the groups.C4 Predicting and identifying reactions and products.C2.3.2 explain how observed simple properties of Groups 1, 7 and 0 depend on the outer shell of electrons of the atoms and predict properties from given trends down the groups.C2.3 How do metals and non-metals combine to form compounds?.6.13 Explain the relative reactivity of the halogens in terms of electronic configurations.6.5 Explain this pattern in reactivity in terms of electronic configurations.5.1 Atomic structure and the periodic table.Explain how the observed simple properties of Group 7 depend on the outer shell of electrons of the atoms and predict properties from given trends down the group.Explain how the observed simple properties of Group 1 depend on the outer shell of electrons of the atoms and predict properties from given trends down the group.Students should be able to: explain how properties of the elements in Group 1 depend on the outer shell of electrons of the atoms.Students should be able to: explain how properties of the elements in Group 7 depend on the outer shell of electrons of the atoms.4.1 Atomic structure and the periodic table.Explanations in terms of atomic radius, screening effect and nuclear charge for general trends in proprties of elements in groups I and VII.Explanations for general trends in values: (i) down a group Explanations for trends in values: (i) down a group The periodic table as a list of elements arranged so as to demonstrate trends in their physical and chemical properties.Unit C1: Structures, Trends, Chemical Reactions, Quantitative Chemistry and Analysis.1.6.20 demonstrate knowledge and understanding of how the reactivity down the group depends on the outer shell electrons of the atoms. 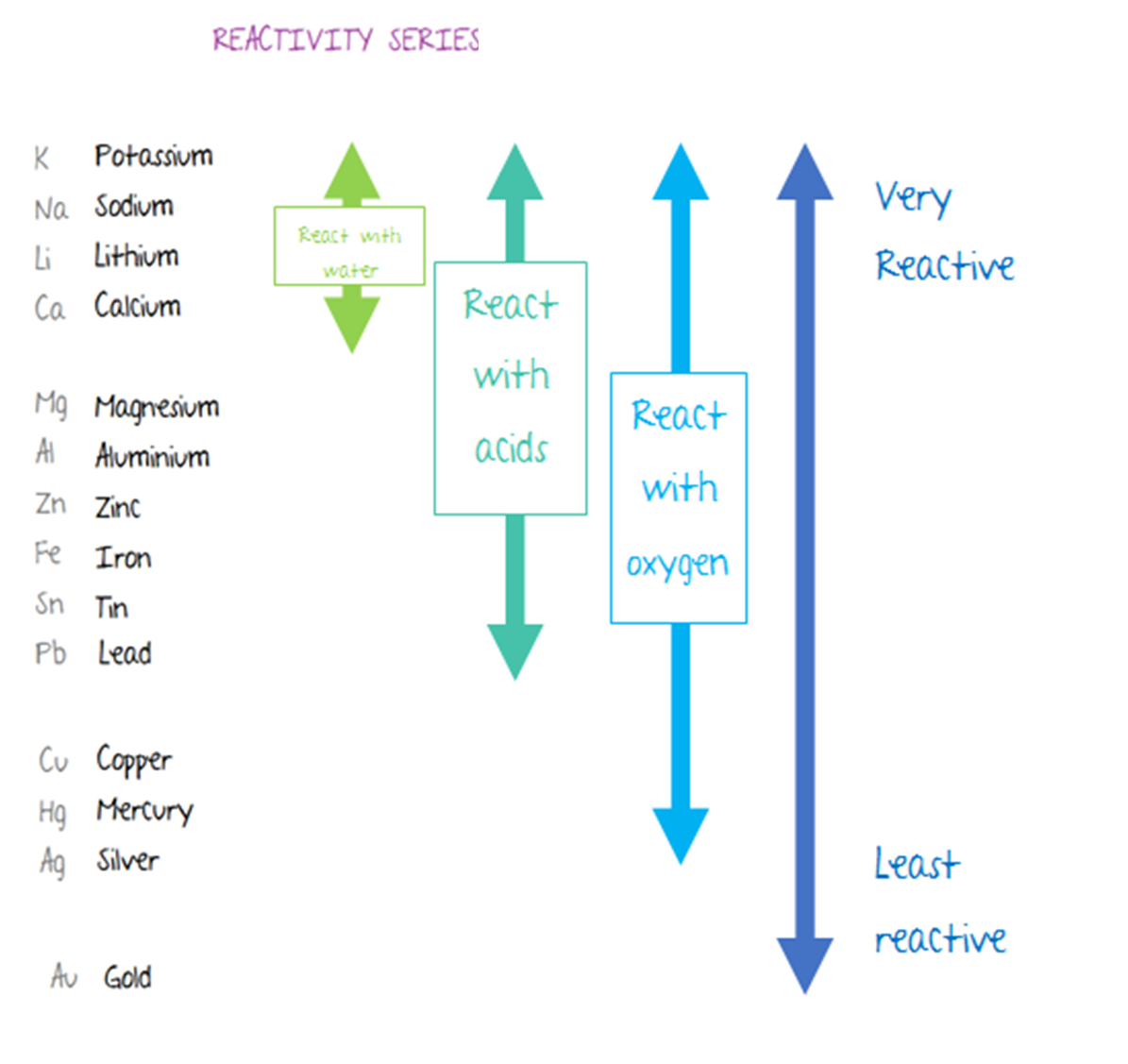
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
